Rust! The eternal enemy of metals. It eats away, corrodes and destroys without mercy. It stops at nothing, not even steel giants like ships and bridges, let alone your garden fence at home. But even when fighting it seems hopeless, there are some ways and means to keep the iron eater at bay! How can rust be removed? How can you prevent rust? Here you’ll get all the answers!
How does rust form?
How rust forms is a long and rather complicated story. I will try to it break down to the essential scenario for you.
Oxygen corrosion
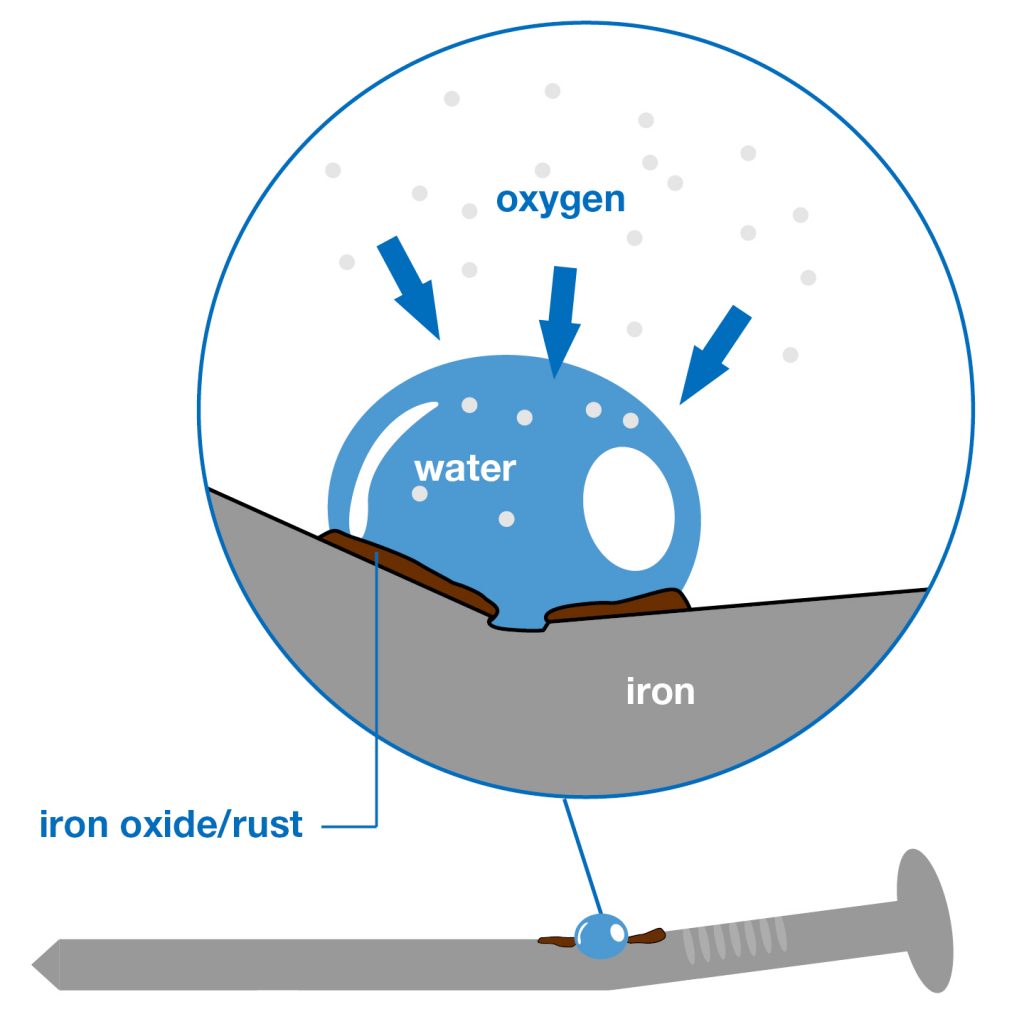
Moisture and oxygen play the leading roles in this process. It all starts with oxygen stealing two electrons from the iron. The oxygen just can’t help itself, as it lacks exactly those two electrons in order to completely fill its outer eletron shell. This “theft“ is called oxidation. However, the iron can deal with oxidation alone and remains unharmed. A thin layer of iron(II) oxide protects the metal against further destruction.
It’s only when water comes into play, too, that the iron is in serious trouble. For then the oxygen bonds to the water molecules instead of the iron. In a desperate attempt to compensate for the loss of electrons in water, the iron gives off atoms to the water. The slow dissolution of the iron is called corrosion. So keep in mind: Rust forms as a result of corrosion and oxidation.
Anti-rust treatment and corrosion protection
Remove or prevent rust?
When fighting rust, one question is essential before you start: Do you want to remove rust or do you want to prevent it in the first place? Depending on how you answer this question, there are different products you can resort to. It’s a little bit like caries. Do you need tooth paste which prevents caries or do you already have a hole in your tooth which needs filling?
Surface protection
- Stainless Steel Spray (also available in bright grade)
- Zinc Spray (also available in bright grade)
- Brass Spray
- Copper Spray
- Brushable Liquid Zinc Paint
- Aluminium Paint
- Copper Paste
What do all these products have in common? They all provide a protective shield and separate the iron from its corrosive environment. How exactly the chemical reactions of the different sprays work, would, however, go beyond the limits of this blog post.
But let’s, for example, take a look at the WEICON Zinc Spray, which consists of microfine zinc flakes. Zinc protects against corrosion in two different ways. For one thing, as already mentioned, the applied layer ensures the separation of steel and its corrosive environment. Secondly, zinc provides another, more active way of corrosion protection. Chemically speaking, zinc is less precious than iron, which is why it gives off its electrons in a moist environment and therefore “sacrifices“ itself for the iron, so to speak. The correct term for this process is redox reaction.
But enough of chemistry for now. You’re probably more interested in how you can tackle rust in daily life.
Remove rust – products for your home
I’m sure many of you have a little rust museum of their own at home. The exhibits often include rusty bike chains, poorly maintained tools, old banisters, or a weathered garden gate. I will now present to you several products, which are able to remove and prevent rust.
Banister
Your old banister keeps rusting? Here’s what you can do about it! First, sand down the banister throughly and clean the surfaces with WEICON Surface Cleaner. Then it’s time for our WEICON Rust Converter. The Rust Converter ensures effective neutralisation of rust and provides short-term protection against corrosion.
Finally, apply the above-mentioned WEICON Zinc Spray to not only give the banister a new shine, but to also to provide it with a long-term protection against corrosion.

Garden gate
If you want to protect your metal garden gate against rust, WEICON Zinc Spray bright grade is a good choice. The weather-resistant spray contains zinc and aluminium pigments with a high level of purity and thus provides excellent corrosion protection.
Tools
Are your garden shears rusty or do you have wrenches covered with rust spots in your toolbox? Then you should resort to our multifunctional oil WEICON W 44 T® Multi-Spray. It does not only protect against corrosion, but also loosens already existing rust due to its excellent capillary action.

Bike chains
You can also use the Multi-Spray to care for your bike chain! The wafer-thin protective film repels water and preserves metal. That makes the bike chain a lot more durable.

Restore bike
Do you have an old, rusty bike in your basement and you’re not sure if you can save it or if it’s ready for the scrap heap? In another blog post, we’ve shown you how to turn an old, worn-out bike into a gorgeous vintage piece. To do so, we used many of our anti-corrosion sprays, like WEICON Rust Protection 2000 PLUS or WEICON Aluminium Spray A-100. Check it out!

Drawbar
The drawbar of a trailer or caravan is prone to rust, if the galvanization is already a few years old. WEICON Corro-Protection – as the name already tells – provides safe corrosion protection for blank, non-varnished metals.

Tow hitch
The ball-shaped head of a tow hitch tends to start rusting, if it isn’t maintained regularly. In this case, WEICON Rust Converter is a handy helper again. Just apply it to the tow hitch and the rust is neutralised in no time.
Our trading partner fighting against rust
In this video, one of our customers demonstrates how to restore rusted metal with our Cleaner Spray S, Rust Converter and Chrome-Silver Spray.
The end of rust – product overview
I’m sure you have all sorts of other things at home which need protection from the eternal enemy of metals. And our numerous products for corrosion protection and anti-rust treatment are likely to be suitable for many of your belongings. For a better overview, here’s a comprehensive summary table for you!
And now let’s get to work! Or how does the saying go? If you rest, you rust!